  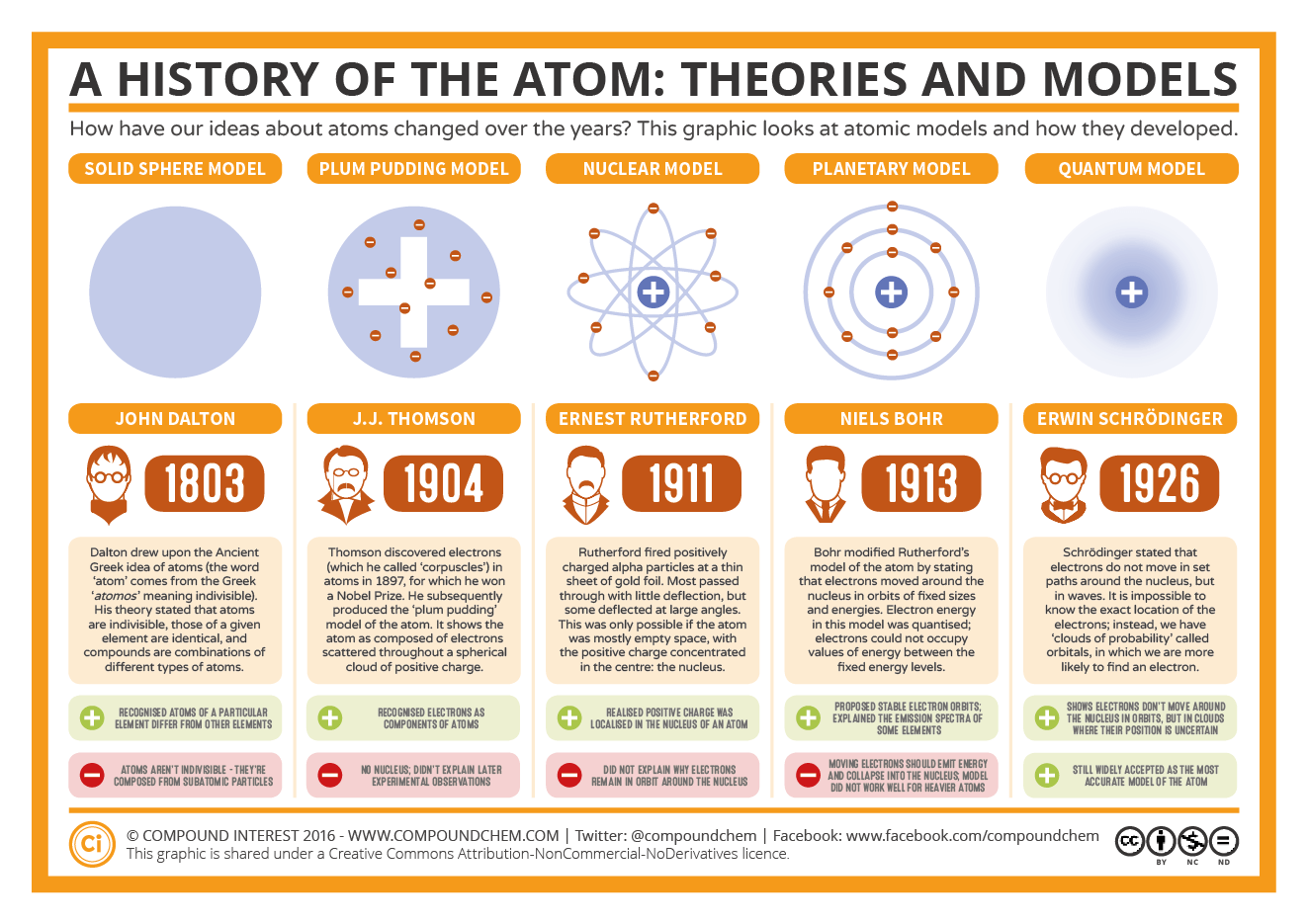
Rutherford hypothesized a planetary model of the atom in which a sphere of negative electrification of charge –Ne (where e is the charge of the electron) surrounded the nucleus of total charge +Ne due to the attraction generated by the Coulomb potential of the nucleus. Rutherford proposed a different model in 1911 after the result of the Geiger-Marsden experiments performed at the Manchester laboratory had convinced him that all the positive charge was concentrated in the pointlike centre of the atom, which he later called the nucleus. In the Thomson model, the negatively charged electrons were the only corpuscular constituents of the atom, while the electrical neutrality was obtained by hypothesizing a substance that surrounded the electrons and whose positive charge perfectly balanced that of the atomic electrons. At that time, electrons were the only subatomic particles whose existence was widely accepted because of various experimental observations, culminating in Thomson’s verification of the constancy of the electron charge-mass ratio in 1897. Thomson had been building the first well-known dynamical model of the atom since 1903. They had also formulated two different models of the atom. The former succeeded Lord Rayleigh as the third director of the Cavendish Laboratory in Cambridge in 1884, while the latter had instituted his laboratory in Manchester in 1907. Both Thomson and Rutherford had established two flourishing schools of experimental physics housed in two different laboratories. Rutherford, who had been awarded the Nobel Prize in Chemistry in 1908 for his studies on radioactivity. Thomson, who had received the Nobel Prize in Physics in 1906 for his discovery of the electron and E. 
The following steps of Bohr’s intellectual life concerned his research in England with two of the most authoritative experimental physicists of the period: J. The problem Bohr underlined in his dissertation was, indeed, resolved only after fundamental developments of quantum theory, such as the formulation of the exclusion principle by Wolfgang Pauli in 1925 and the independent development by Enrico Fermi and Paul Dirac of the statistics of the particles obeying said principle in 1926. This conclusion was fundamental in giving Bohr the conviction that a revision of classic electromagnetism was necessary, in order to deal with atomic phenomena. By generalizing the assumptions of the Lorentz-Drude theory, Bohr deduced that it was not possible to derive the diamagnetic and paramagnetic properties of metals from the accepted laws of electromagnetism. The Lorentz-Drude theory explained some of the electrical and thermal properties of metals, but several experiments disagreed with the values predicted by the theory. The theory on which Bohr based his study was the Lorentz-Drude model, according to which metals were depicted as gases of electrons moving almost freely in a potential generated by positive charged ions fixed in a crystal structure. The mixture of theoretical ability and physical intuition that Bohr showed in handling this problem remained one the central features of Bohr’s style.īohr began his epoch-making study of the structure of matter with his master’s thesis on the electron theory of metals - a topic that he further elaborated in his PhD dissertation completed in 1911. 
To deal with this question, Bohr had to deepen both the experimental and theoretical issues concerning such a calculation. Although Bohr is remembered for his fundamental contributions to theoretical physics, his first work concerned the purely experimental question of measuring the surface tension of a jet of liquid emerging from a cylindrical tube. He began his studies in physics in 1903 at the University of Copenhagen, then the only university in Denmark. Even before becoming a university student, Bohr attended gatherings with important exponents of the Danish intelligentsia, such as the philosopher Harald Høffding, who had a long-lasting influence on the philosophical outlook which Niels Bohr brought to physical problems. Since his boyhood, Bohr enjoyed the lively cultural environment surrounding his father’s professional life. "for his services in the investigation of the structure of atoms and of the radiation emanating from them".īorn in Copenhagen on October 7 1885, Niels Bohr was the second of three children of the eminent Danish physiologist Christian Bohr. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
